- Name:Venturepharm (Haian) Co., Ltd.
- Category:Corporation/Limited Liability Company
- Trade Category: Manufacturer
- Employees:101 - 500 People
- Address:Nanhai Road 279, Xiuying District, Haikou City
- Website:
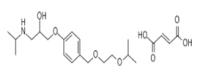
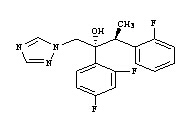
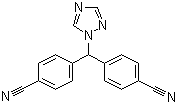
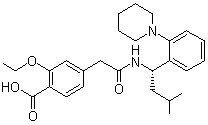
- Product/Service:Voriconazole, Bisoprolol, Adapalene, Loperamide, Fexofenadine, Riluzole, Letrozole
VP API is one of the four fonctional management parts of Venturepharm Group. Mainly focus on the providing of contract service, technical support of new products, rare APIs production, consultant to t...
View More >


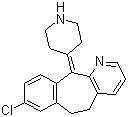
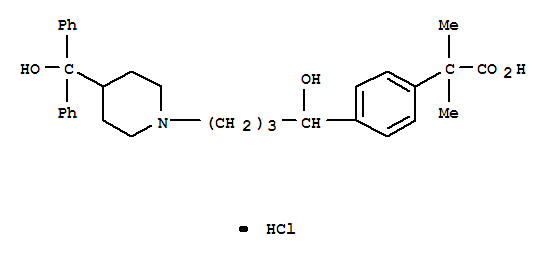
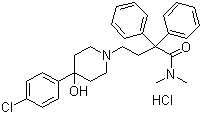
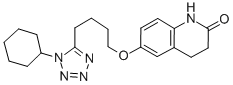
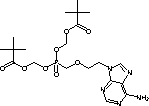
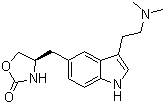
 Info marked by "
Info marked by " Venturepharm (Haian) Co., Ltd.
Venturepharm (Haian) Co., Ltd.